 How would I find the value of t What is a possible drawback to direct democracy ? How might it be solved ? What is. A: metabolismĭ: calorie What is the value of the expression 9 x How many water molecules are in a block of ice containing 1.75 mol of water (H2O)? What is the difference between Sargon and Hammurabi What’s the plural form of wheat Most engines will contain how many cylinders in order to maintain a proper balance of weight and forces? A. Shakespears use of more than 10 syllablesbper line Express the following number using expanded form, unit form, and word form: 625.25 Which of the following are true about alloy steel chains ? Explain how you can find 4 × 754 using two different methods What is the cube root of 4096 What is the area of a circular mirror with a radius of 10 cm? A _ is a substance the body needs in order to grow and function properly. Rank the compounds in order from largest mass percent of sodium to smallest mass percent of sodium. How much more time did Dan spend on his math assignment? Using what you know about the compressibility of gases explain why the oxygen in is a SCUBA tank is compressed Sodium hydroxide, NaOH sodium phosphate, Na3PO4 and sodium nitrate, NaNO3, are all common chemicals used in cleanser formulation. then he completed his chemistry assignment in 1/4 hours. Which expression represents Bella's score on a spelling test if she studies for 3 hours? A.ē(score)ĭ.ē - score Aaron finished his math assignment in 3/8 hours. Why do you think dystopian literature has been so common throughout history? Bella's score on a spelling test is a function of the number of hours she studies. To find more about compounds, refer the link below: Hence, the two compounds in the given lists are sodium chloride and carbon dioxide.Thus, option D is correct. Similarly carbon dioxide is a covalent compound formed from carbon and oxygen atoms. 
sodium chloride (NaCl) os an ionic compound of sodium and chlorine. The compounds are classified based on their bond type, composition etc. There are different types of compounds such as ionic compounds, covalent compounds, coordination compounds etc. Sodium, iron, aluminum ets are single atoms and not compounds. For example water is a compound formed from two hydrogens and one oxygen atom.Ĭombination of same atoms forms molecules but not compounds. The two compounds in the given lists are sodium chloride and carbon dioxide.Ĭompounds are chemical substances formed by the combination of two or more different elements. Sodium has many isotopes, out of which only one isotope is most stable (i.e 23Na).Each elements, molecules and compounds have their own formula to identify them. It is always found in a compound form with other elements. In nature, Sodium never exists as a pure metal because of its high reactivity.Out of all alkali metals, Sodium is most abundant. Around 2.6% of the Earth’s crust contains sodium metal.Sodium element is also essential to maintain the fluid balance in the cells of our body.Sodium burns with a golden yellow flame when it is heated.Sodium metal is lighter than water, hence it floats on water.Sodium metal is silver white in color when it is freshly cut, but it suddenly forms an oxide layer if kept open in air.Sodium is a soft metal and it can be cut even by using a kitchen knife. 
Here are the interesting facts about Sodium element. That means sodium metal is less dense than water.īecause of this reason, the sodium metal floats on water. Sodium has a density of 0.97 g/cm 3, while water has a density of 1 g/cm 3. Now let’s see about sodium metal and it’s density. And if the density of object is less than the water, then it will float on water. If the object has a density higher than the water, then it will sink. You might have studied the concept of density in physics.īy knowing the density of two objects, we can come to know which object will float and which will sink in water. 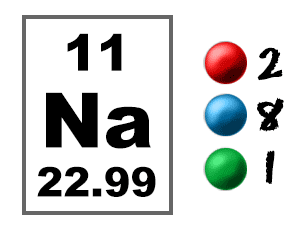
Silvery white State (at STP) Solid Position in periodic tableĪlkali metals Atomic number or Protons 11 Neutrons 12 Electrons 11 Symbol Na Atomic massĢ, 8, 1 Electronic configuration 3s 1 Atomic radiusĢ27 picometers (van der Waals radius) Valence electronsġ 1st Ionization energy 5.139 eV ElectronegativityīCC (Body Centered Cubic) Melting point 370.9 K or 97.7 ☌ or 208 ☏ Boiling point 1156 K or 882.9 ☌ or 1621.2 ☏ Density 0.97 g/cm 3 Main isotope 23Na Who discovered Sodium and when? Sodium Element (Na) Information Appearance So if you want to know anything about Sodium element, then this guide is for you. In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Sodium element in Periodic table.) The electron configurations and orbital diagrams of these four elements are: The alkali metal sodium (atomic number 11) has one more electron than the neon atom. This is a SUPER easy guide on sodium element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
